Proteins are commonly built up by folded domains connected by regions with higher flexibility.
The interdomain orientations encoded by such hinges or linkers can play central roles in controlling
the function of multidomain proteins, which makes them important to characterize.
Small Angle X-ray Scattering (SAXS) is uniquely suited to study the conformational ensembles adopted
by these kinds of proteins. However, because of the limited information provided by SAXS, ensemble
models must be built by combination with other information sources and care have to be taken to avoid
constructing ensembles that are more complex than data can support.
We developed a method based on Bayesian statistics that combine data from molecular simulation with
experimental data from SAXS and Nuclear Magnetic Resonance while automatically balancing the
complexity of ensemble model with information in the data. We demonstrate that this method is capable
of accurate inference of ensembles even in the presence of high levels of experimental noise.
The method represents a general approach to combine data and simulation in the modeling of
protein ensembles and can be extended to employ additional sources of experimental information.
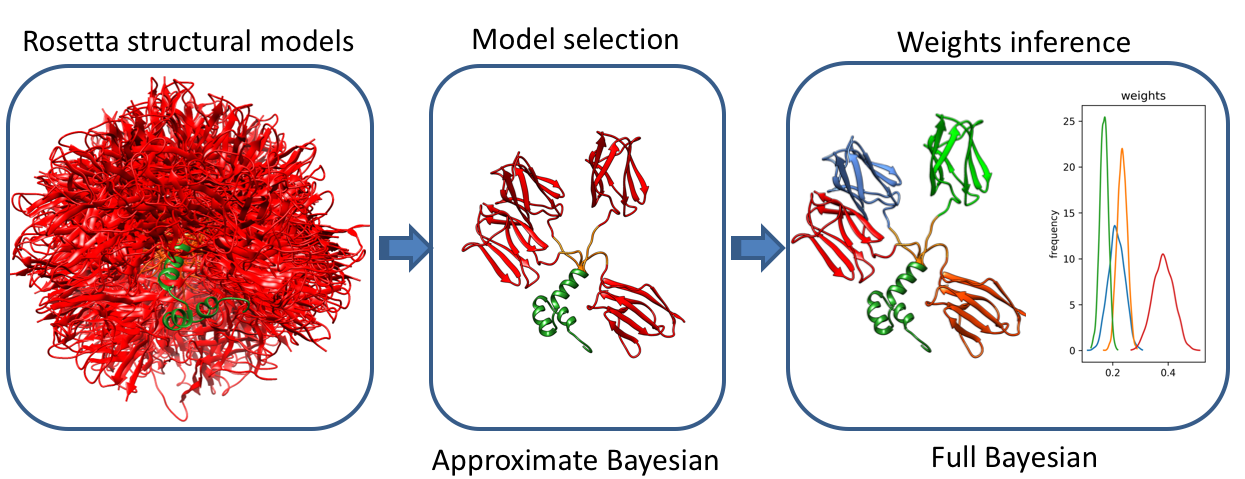
Bioce is a method based on Bayesian statistics that infers conformational ensembles
from a structural library and experimental Small Angle Scattering data (SAXS or SANS).
The first stage of the method involves a fast model selection approach based on variational Bayesian
inference that maximizes the model evidence of the selected ensemble.
This is followed by a complete Bayesian inference of population weights in the selected ensemble.
More details about the method can be found in: Citation: Potrzebowski W, Trewhella J, Andre I (2018)
Bayesian inference of protein conformational ensembles from limited structural data.
PLoS Comput Biol 14(12): e1006641. https://doi.org/10.1371/journal.pcbi.1006641
https://doi.org/10.1371/journal.pcbi.1006641